What Type of Intermolecular Forces does Isopropanol have?
Isopropanol has hydrogen bonding, dipole-dipole, and London dispersion. Hydrogen bonding is bonds between hydrogen and either fluorine, oxygen, or nitrogen. Dipole-dipole bonding is when there is bonding between molecules because the positive element of one molecule bonds with negative element of the other molecule. Lastly, London dispersion is when bonding acts on two molecules because they are adjacent to each other. The three molecules I chose to bond with isopropanol are isopropanol, water, and ethanol. I chose these three because they are polar and that means that they can bond with the polar molecule of isopropanol.
Examples of Bonding...
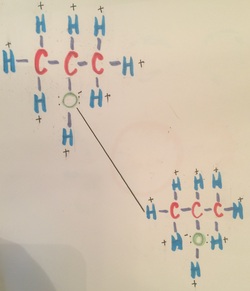
This drawing is showing isopropanol bonding with isopropanol. This bond has three forces involved with the bonding. Force one is hydrogen bonding. You can tell it is hydrogen bonding because the oxygen of one isopropanol model is bonding with the hydrogen in the other isopropanol model. Another bond is dipole-dipole. This is because the negative from the oxygen is bonding with positive of the hydrogen. Lastly it is considered London dispersion because it acts only two molecules that are adjacent to each other.
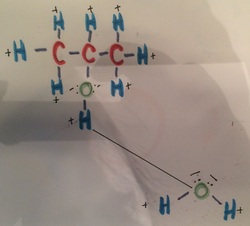
This is drawing showing isopropanol bonding with water. This bond also has three forces involed with it. With this hydrogen bond, the hydrogen from the isopropanol model is bonding with oxygen from the water molecule. This dipole-dipole bonding is showing the positive hydrogen bonding with the negative from the oxygen. Lastly, London dispersion is bonding the isopropanol and water because they are adjacent to each other.
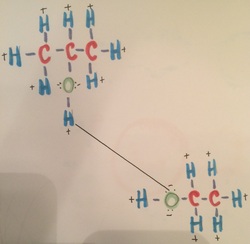
This picture is showing isopropanol bonding with ethanol. This bonding has all three of the forces as well. The hydrogen bonding is between the hydrogen of the isopropanol and the oxygen of the ethanol. The dipole-dipole is happening because the negative from the oxygen in the ethanol molecule is bonding with the positive from the hydrogen in the isopropanol molecule. Lastly, the London dispersions acting on the two molecules because they are adjacent to each other.
